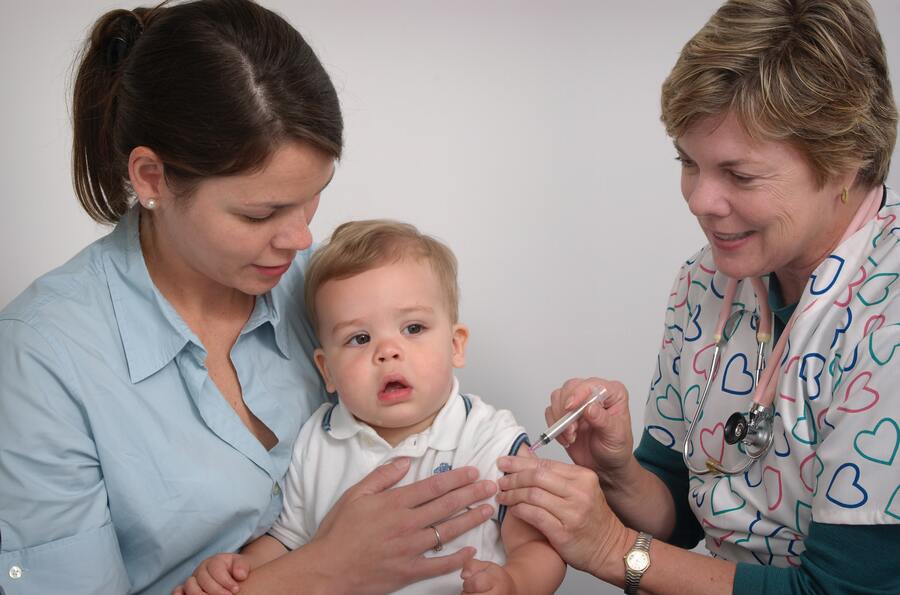
News broke just last night as Pfizer and BioNTech officially announced a submission to the U.S. FDA for administering the branded vaccine to children under 12 years old.
This follows a clinical trial period across some 4,500 children from the age of six months to 12 years from over 90 cities in the U.S., Finland, Poland, and Spain.
According to their forward-looking reports, in that data derived is applied to projection, a favourable profile is to be expected, with robust neutralising antibody responses noted. The children underwent a two-dose regimen of 10µg Pfizer-BioNTech doses over a 21-day gap schedule. A previous study conducted with 30μg doses for those aged 16 to 25 reported similar favourable results, with the dosage adjusted in consideration of safety, tolerability, and immunogenicity of the respective age ranges.
The U.S. FDA has previously issued a warning of rare heart inflammation risk for children who receive the Pfizer-BioNTech vaccine.
These new findings indicate a large-scale inoculation for the world, welcoming the next level of the vaccination programme — the prevention of Covid-19 contagion among children.
Malaysia aims for the end of 2021 or the beginning of 2022’s school terms to achieve a majority of kids receiving at least partial vaccinations. Current Minister of Higher Education Datuk Seri Noraini Ahmad stresses the need for vaccination roll-outs and registration, already passing campus regulations for on-site attendance. Tertiary level students are required to provide proof of receiving both vaccinations to attend university and college, with supplemental classes to be provided for those still partially vaccinated. This is in line with the IPT Recovery Plan (Plan Pemulih IPT) due to commence officially in October.
As of now, only the Pfizer-BioNTech vaccine has been approved by Malaysia’s National Pharmaceutical Regulatory Agency (NPRA) for children aged 12 and up. The Health Ministry targets a 60% partial vaccination of Malaysia’s 12- to 17-year-olds by November.
More data from the vaccine companies are still awaiting submission for any clarity on immunisation plans for under-12s.
Conversely, China has already begun vaccinating their 3- to 17-year-olds with the Sinovac vaccine, one of the first countries to reach the younger demographic.
"ExpatGo welcomes and encourages comments, input, and divergent opinions. However, we kindly request that you use suitable language in your comments, and refrain from any sort of personal attack, hate speech, or disparaging rhetoric. Comments not in line with this are subject to removal from the site. "